Desulfurization process application
|
Desulfurization Scheme |
Main Process |
Process Characteristics |
| Wet Flue Gas Desulfurization (WFGD) Technology | Limestone wet method, magnesium method, ammonia method, double alkali method | Fast desulfurization reaction rate Desulfurization absorption is carried out under low temperature and wet saturated conditions; High desulfurization efficiency However, the system is more complex, the construction and operation and maintenance costs are high, and environmental problems such as wastewater and white smoke are generated. |
| Dry Flue Gas Desulfurization (DFGD) Technology | In-furnace calcium injection/post-furnace humidification and activation desulfurization technology/SDS | Solid absorbent reacts with SO2 in a dry state; no secondary pollution from waste liquid. However, the reaction rate is slow, and the desulfurization efficiency and desulfurizer utilization rate are low 。 |
| Semi-dry Flue Gas Desulfurization (SDFGD) Technology | Rotary spray drying process, circulating fluidized bed desulfurization process | Desulfurizing agents generally desulfurize in a wet state, with dry by-products; semi-dry processes have some characteristics of both WFGD and DFGD technologies |
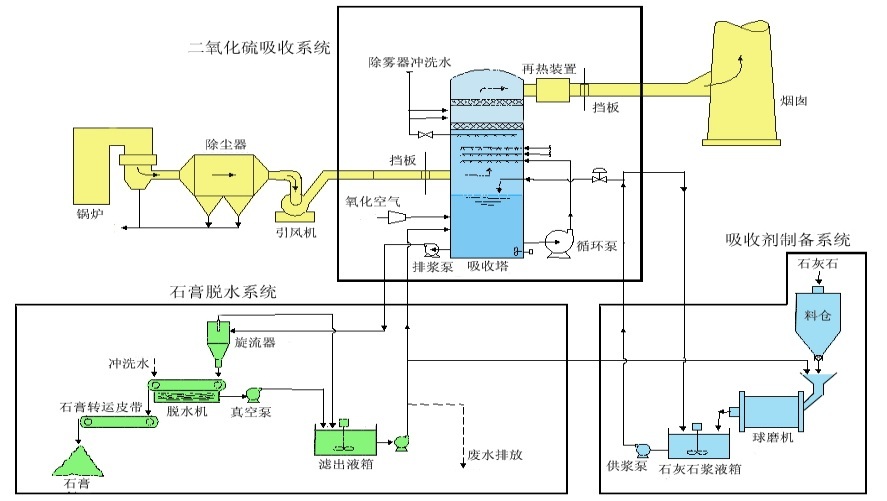
1. Limestone-gypsum wet desulfurization technology
(1) Reaction mechanism and process flow
Reaction mechanism:
Limestone decomposition
CaCO3 (s) ---> CaCO3 (aq)
CaCO3 (aq) + H2O ---> Ca2+ + HCO3- + OH-
SO2 absorption
SO2 (g) ---> SO2 (aq)
SO2 (aq) + H2O ---> H2SO3 ---> HSO- + H+ ---> H+ + SO32-
Forced oxidation in the absorption tower
SO32- + 1/2 O2 ---> SO42-
The basic reactions in the absorption tower are as follows:
SO2 + CaCO3 + 1/2O2 ---> CaSO4 + CO2
Other side reactions:
Ca2+ + SO32- + 1/2 H2O ---> CaSO3.1/2H2O(s)
2 HCl + CaCO3 ---> CaCl2 + H2O + CO2
2 HF + CaCO3 ---> CaF2 + H2O + CO2
(2) System composition
|
▷ Limestone slurry preparation system |
 |
(3) Unique technology
|
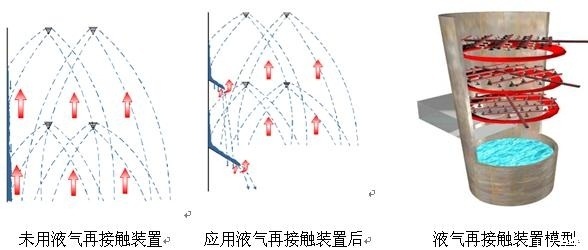
Gas-liquid Recontaction Device |
|
|
Gas distribution device |
|
Tray
1) The slurry sprayed from the high-efficiency nozzle uniformly falls onto the tray, and under the action of the liquid holding layer above the tray from the evenly distributed flue gas entering the absorption tower, the SO2 in the flue gas is fully washed and removed.
2) The tray extends the residence time of flue gas in the absorption tower, and utilizes the condition that the liquid film PH value is lower than that of the slurry pool to promote the dissolution of limestone, thereby improving desulfurization efficiency and reducing the gas-liquid ratio.
High-efficiency nozzle
With the same flow rate, a 120° high-efficiency dual-head nozzle is used, resulting in smaller slurry particle size and higher coverage. The high-efficiency dual-head nozzle can secondary atomize the droplets, dispersing the droplets into smaller particle sizes, increasing the specific surface area of dust removal, and improving dust removal performance and desulfurization efficiency.
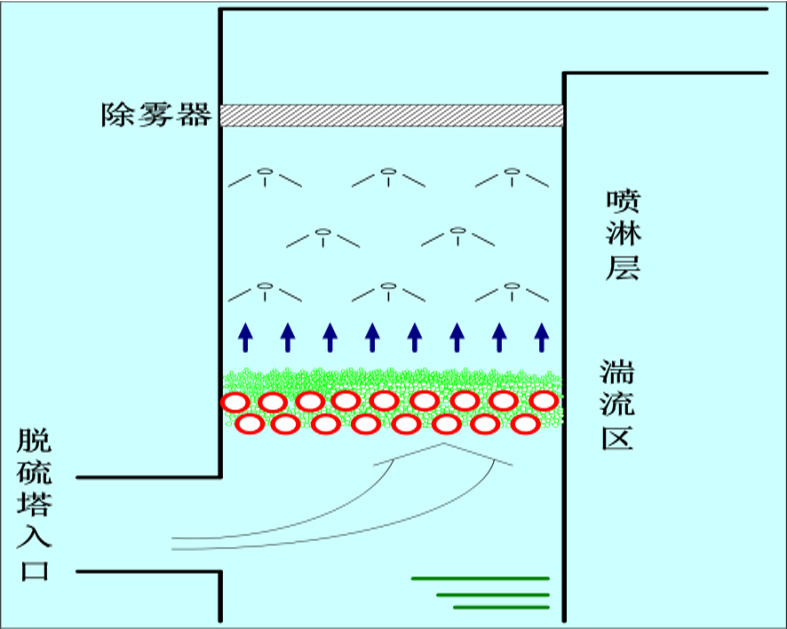
High-efficiency demister
Using a three-stage ridged high-efficiency demister and increasing the distance between the topmost spray layer and the demister can control the outlet mist content to below 20 mg/Nm3, thereby reducing the amount of dust carried by the mist droplets and meeting the ultra-low emission requirement of 5 mg/Nm3.

4) Technical Features
◆The absorption tower is a spray empty tower, with simple operation and high reliability.
◆The absorption tower is designed for high flue gas flow rate (3.4-3.8 m/s), good mass transfer, and small footprint.
◆Low gas-liquid ratio, low power consumption, and low operating cost.
◆The slurry uses multi-layer countercurrent spraying, with a spraying coverage rate of 300%, and a desulfurization efficiency of over 99%.
◆The nozzle is a hollow cone type, convenient for maintenance, and not easily blocked.
◆An eaves is set at the absorption tower inlet to prevent slurry from entering the flue, avoiding scaling.
◆In-tower oxidation is adopted, and the obtained gypsum particles have uniform diameter, which is convenient for gypsum dehydration.
◆A gas-liquid re-contact device is set between the spray layers to prevent flue gas from escaping.
◆Multiple pollution control can be achieved, and the dust removal efficiency can reach 50-70% while desulfurization.
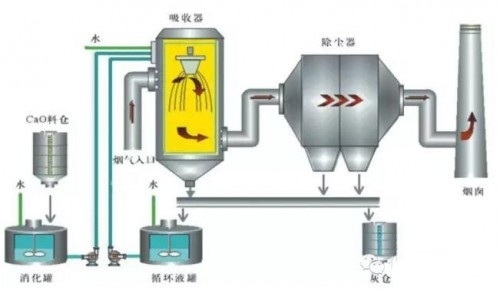
2. CFB Circulating Fluidized Bed Semi-dry Desulfurization Technology
1) Reaction Mechanism and Process Flow
CaO + H2O → Ca(OH)2 (1)
SO2 + H2O → H2SO3 (2)
Ca(OH)2 + H2SO3 → CaSO3 + 2H2O (3)
CaSO3 (L) → CaSO3 (S) (4)
Based on the circulating fluidized bed principle, the contact time between the absorbent and the flue gas is extended through multiple recirculation of the absorbent, and the utilization rate of the absorbent is improved. After passing through the Venturi tube at the bottom of the absorption tower, the flue gas is accelerated to obtain the power to form a fluidized bed. After entering the bottom diffusion section, the flue gas speed decreases, and a fluidized bed layer is formed here, which is also the main area for desulfurization chemical reactions. Above the fluidized bed layer is the straight pipe section of the absorption tower, which can be divided into: dense phase zone, transition zone, and dilute phase zone. After passing through the "transition zone", the desulfurization chemical reaction has basically ended. The temperature and pressure are detected in the top turning section of the absorption tower. The water addition amount of the absorption tower is controlled by the temperature, and the desulfurization ash circulation amount is controlled by the inlet and outlet pressure drop of the absorption tower.
|
2) System Composition |
|
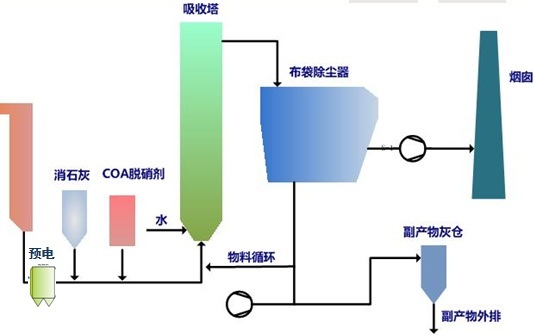
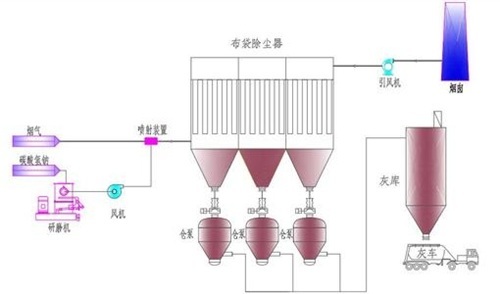
3. SDA Desulfurization System Characteristics
SDA utilizes the principle of spray drying. After the wet absorbent is sprayed into the absorption tower, it absorbs sulfur dioxide from the flue gas through a chemical reaction. Simultaneously, the flue gas transfers heat to the absorbent, causing it to dry continuously. The desulfurization reaction's waste residue is discharged from the conical outlet of the absorption tower in dry form, thus called semi-dry flue gas desulfurization.
|
1) Process Flow 2) Desulfurization System Characteristics |
|
|
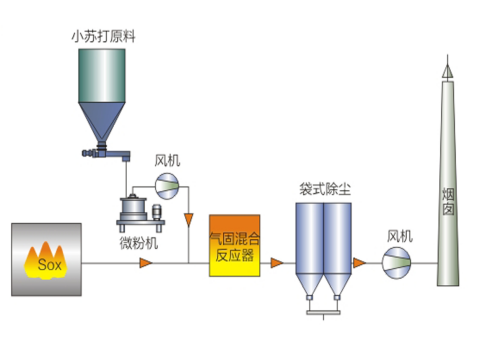
4. SDS Desulfurization System Characteristics 1) Reaction Mechanism and Process Flow The main chemical reactions are: NaHCO3 +HF → NaF +CO2+H2O 1) SDS Dry Desulfurization Technology Characteristics |
|
No. 169, Jingwu Road, Lixia District, Jinan, Shandong Province, China
Service Hotline

Follow Ruilong Environmental Protection